4-Hydroxynonenal is both a substrate and an inhibitor of ALDH2; inhibition of ALDH2 by 4-Hydroxynonenal is reversible at low concentration and become irreversible when the concentration of 4-HNE reaches 10 μM.
4-Hydroxynonenal can induce antioxidant defense mechanisms to restrain its own production and to enhance the cellular protection against oxidative stress.
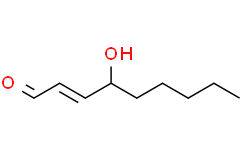
4-Hydroxynonenal, the product of lipid peroxidation, is mutagenic and genotoxic in viruses, bacteria and mammalian cells. It reacts with all four DNA bases but with different efficiency: G >C > A >T. 4-Hydroxynonenal-dG represents the best biomarker of the genotoxic effects of 4-Hydroxynonenal and these adducts are primarily found in nuclear DNA. A classic example of etiological relevance of 4-Hydroxynonenal-dG in human cancers is 4-Hydroxynonenal-dG induced p53 mutation. 4-Hydroxynonenal-dG adducts were preferentially formed at the third base of codon 249 in the p53 gene, causing gene mutation and affecting diverse biological processes including cell cycle arrest, apoptosis, DNA repair, and differentiation.
Medlife has not independently confirmed the accuracy of these methods. They are for reference only.
 扫码关注公众号
扫码关注公众号